Life Sciences
Compliance Starts with the Right Controls
Purpose-Built for Warehouse Management Compliance and Confidence.
Life Sciences Warehousing Capabilities
Our solution
Whether you’re managing pharmaceutical storage, controlled substances, or ultra-low temp inventory, compliance is non-negotiable. Footprint® WMS gives complex life sciences supply chains the tools to meet DSCSA, cGMP, and 21 CFR Part 11 standards—without slowing operations. From nested serialization to validated workflows, Datex helps you maintain full visibility and compliance from receipt to release.
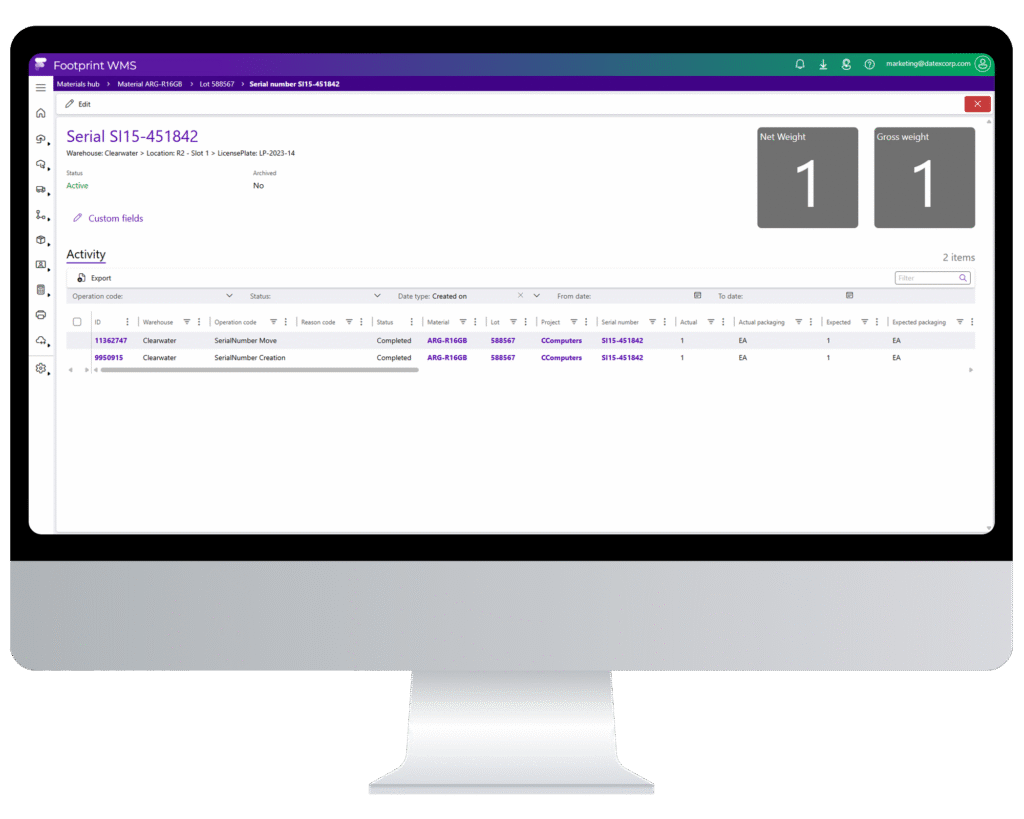
Track & Trace
Compliance Without Compromise
Maintain complete lot, serial, and batch visibility from receipt to shipment with audit-ready traceability that satisfies DSCSA, FDA, cGMP, and 21 CFR Part 11. Whether you’re preparing for an inspection or managing a recall, every movement, user action, and timestamp is digitally logged and instantly accessible.
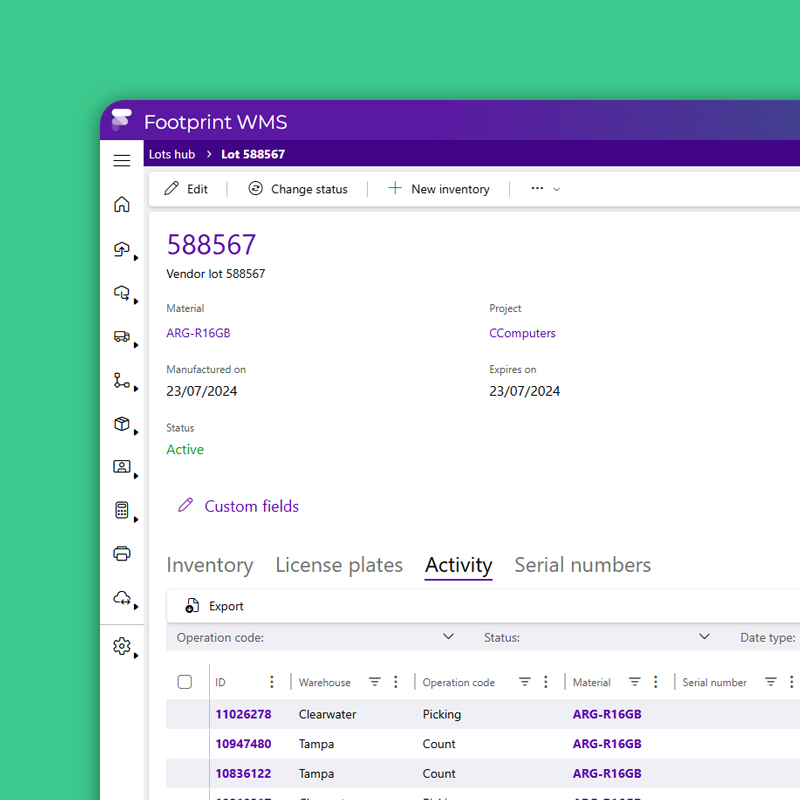
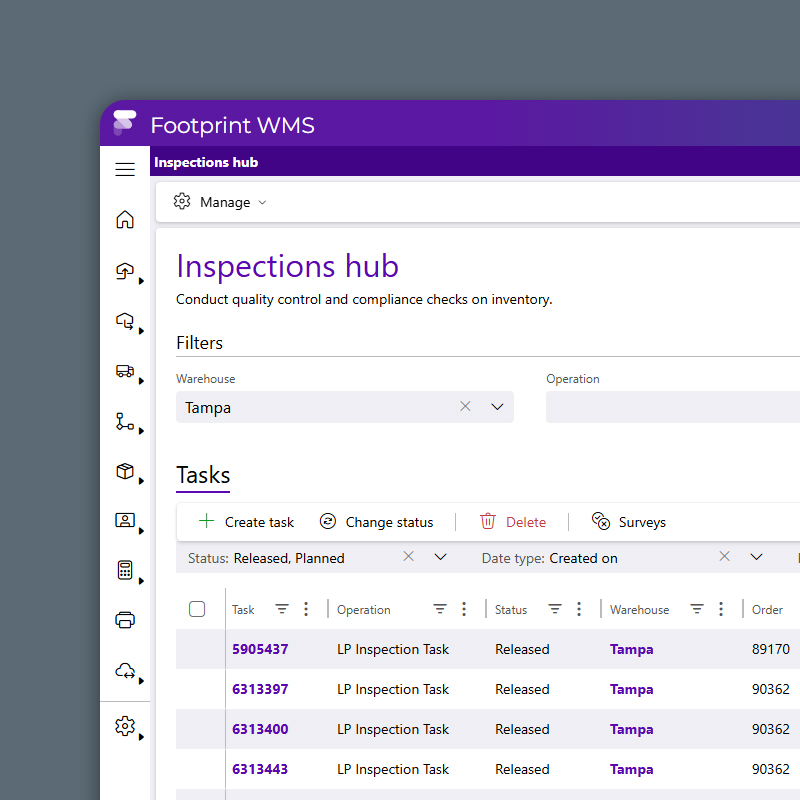
Quality Control
Hold and Release Without the Headaches
Automate QA and QC workflows with rule-based quarantine, photo capture, and inspection logging that adapts to your validation protocol. Batch release doesn’t have to slow down production because Datex Footprint WMS routes tasks by product type, lab result or regulatory requirement to ensure speed and compliance.
Labor Optimization
Productive, Compliant, and Always Aligned
Keep associates on task with workflows that align directly to GxP-compliant procedures. Tailored interfaces reduce risk of human error, while detailed activity logs and role-based access controls ensure validation integrity without adding unnecessary complexity to daily operations.
Discover more on labor optimization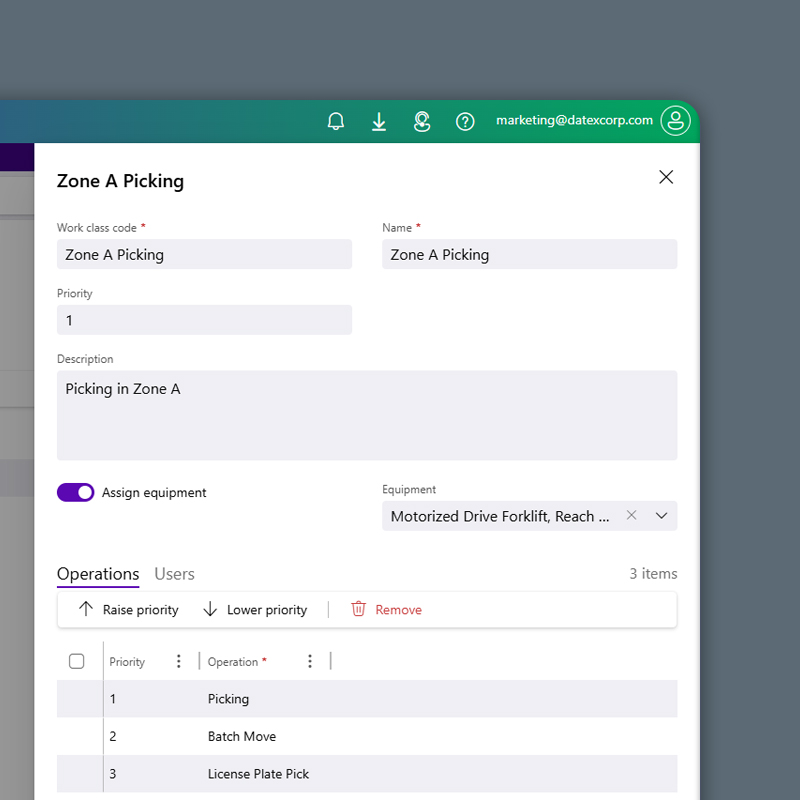
Data Management
Built-In Validation For Built-In Peace of Mind
Datex Footprint WMS provides full support for IQ/OQ/PQ documentation and system validation workflows. With integrations to LIMS, MES, ERP and temperature monitors, your data stays centralized, secure, and audit-ready.
Tobin Scientific: A Precision Partner in Life Sciences Logistics
Case studies
Tobin Scientific provides ultra-low temperature storage and transport for pharmaceutical and research organizations.
They rely on Datex Footprint® WMS to manage strict chain-of-custody requirements, enable digital batch release workflows, and maintain full lot traceability for sensitive, high-value inventory.
Integrations
Seamless third-party integrations to keep your entire business connected.
Leverage Expert Insights and Practical Tools
Industry insights
See what’s going on in the supply chain and logistics industry.
Warehousing Watch: Insights & Innovation | Volume 11
Volume 11 of Warehousing Watch shares practical insights to help life sciences and pharma teams reduce compliance risk, improve visibility,…
Warehousing Watch: Insights & Innovation | Volume 10
Volume 10 of the Warehousing Watch Newsletter: Explore strategies for 3PL longevity, next‑generation labor intelligence, the…
What 3PLs Should Look for in a WMS: An Executive’s Guide to Choosing the Right Partner
Explore the five nonnegotiable capabilities a WMS must have to drive success for a modern 3PL,…
3PL Client Portal Analytics: What Your Customers Actually Want to See
Ditch the manual reports. A 3PL client portal offers the real-time transparency and self-service your customers…
