3PL Life Sciences / Pharmaceuticals
Meet Regulations. Exceed Expectations.
Secure, compliant, and flexible, Datex Footprint WMS gives life sciences 3PLs full control over complex workflows, serialization, and customer-specific requirements at every stage of the supply chain.
Serialization. Security. Scalability. All Solved.
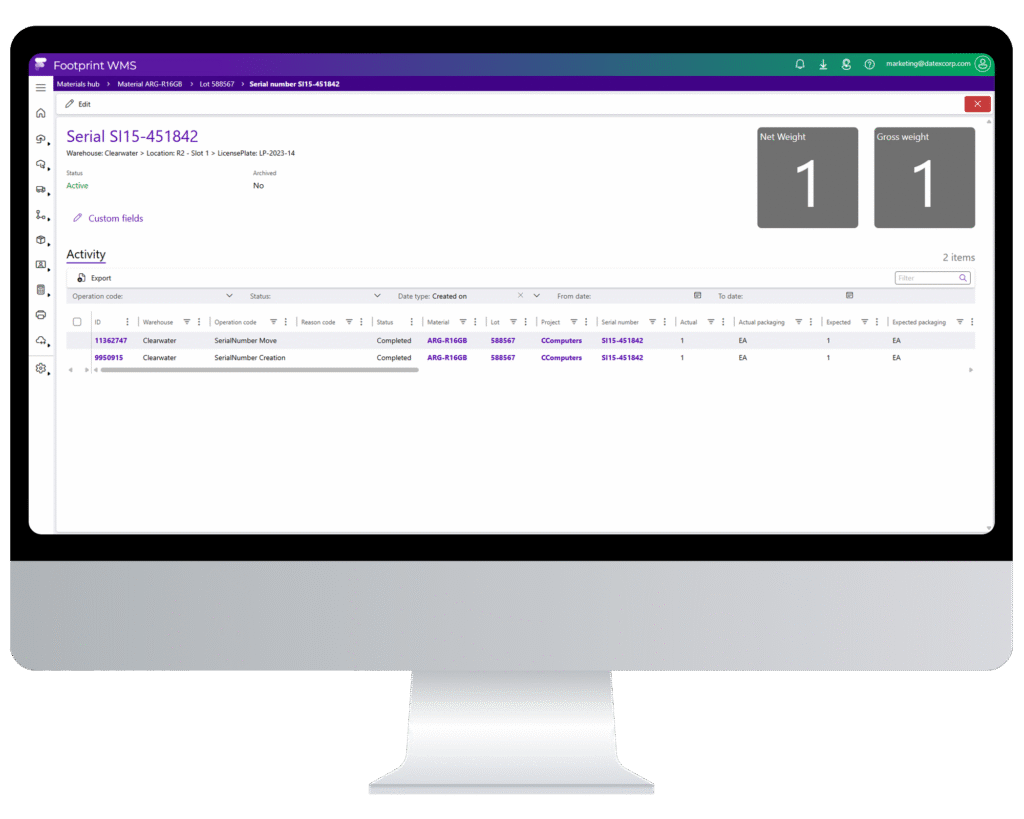
Serialization, DSCSA, and lot-level traceability are now table stakes for your operations. Footprint WMS stands apart by delivering business-critical functionality along with visibility, speed, and configurability across every client, product and process.
Check Out the Mini-GuideSerialization Ready
Track products with full traceability down to the individual unit.
DSCSA Compliance
Aggregate and report GTIN, serial, expiration, and lot details instantly.
Streamlined Validation
Simplify IQ/OQ and docs to meet strict regulatory requirements faster.
Audit & Access Tracking
Record system access and video at key control points for compliance.

David Hamilton
Vice President of cGMP Warehousing, Tobin Scientific
Our pharmaceutical clients need to have a high degree of traceability of the materials going into all of their manufactured batches as government regulations require that level of genealogical data. Datex met that standard.
Traceability and Flexibility. No Tradeoffs.
Datex simplifies compliance with built-in DSCSA, 21 CFR Part 11, and full serialization support from day one. Footprint WMS adapts to client-specific workflows, integrates easily, and delivers the traceability life sciences clients require.
Industry Compliance Without Complexity
- DSCSA, CFR 21 Part 11, and HIPAA Compliant
- Support for Controlled Substances and DEA reporting
- End-to-end serialization aggregation and tracking
Flexible for Every Client, Every SKU
- Configure handling rules per client or product
- Automate labeling, shipping, and documentation workflows
- Support specialty packaging, cold chain, and palletization logic
Integrate Today. Evolve Tomorrow.
- Seamless APIs for ERP, robotics, and other critical systems
- Integrate with third-party serialization and VRS platforms
- Extend functionality easily through Datex Studio
Tobin Scientific: A Precision Partner in Life Sciences Logistics
Case studies
Tobin Scientific provides ultra-low temperature storage and transport for pharmaceutical and research organizations.
They rely on Footprint® WMS to manage strict chain-of-custody requirements, enable digital batch release workflows, and maintain full lot traceability for sensitive, high-value inventory.
